Most lessons learned systems fail for a simple reason: they rely on people to remember, communicate, and follow through. Here is how it usually goes: you become aware of a lesson learned and are tasked with sharing it across different areas, manufacturing lines, and facilities in your organization. You probably schedule some meetings, send a few emails, document the lesson learned somewhere in a spread sheet or database, issue a quality alert, and hope that someone follows through.
You rely entirely on the people you talked to or emailed maybe once or twice, to follow through with the read-across; and when a system depends on people, its outcome cannot be predicted.
The good news is, your organization already has the processes and tools in place you need to ensure robust “read across” or Yokoten. They may need improvement or adjustments, but they exist. I’m referring to the Failure Modes and Effects Analysis tool and the Internal Audit Program – specifically, manufacturing process audits.
A Failure Mode and Effects Analysis (FMEA) is a team-oriented analysis to evaluate the potential risk of failures, their causes and effects. This analysis helps determine and document existing or recommended prevention and detection controls to lower that risk. There are many known and stated benefits of FMEAs. One of the most valuable and often overlooked is how strongly they enable the read-across of lessons learned systematically.
When structured properly, your FMEAs in combination with your internal audit program can provide you with the identification, documentation, communication, and even verification of read across, of a lesson learned. There is no need to add or create a separate process for lessons learned, or a database people may or may not use, or extra meetings and increased follow-ups just to ensure read across. Under this scenario, you will be taking advantage of your established and required quality management system processes to accomplish read across.
For this to work, you will need to structure your failure modes and effects analyses in a hierarchical manner, starting by creating a foundation FMEA atop of the hierarchy. Then, you set-up another FMEA – a family FMEA as a child to that foundation FMEA. The family FMEA will “inherit” (i.e. copy) all content of the foundation FMEA and will contain additional information specific to a family of products. Finally, you will create a product FMEA (if required), under the family FMEA and as a grandchild to the foundation FMEA. The product FMEA will inherit all content from the family FMEA, and therefore the foundation, and will contain additional information specific to that product.
Although these are optional practices in the AIAG|VDA FMEA Handbook 1st (that may be required by some customers), the aforementioned structure, illustrated below, is key for the FMEAs to facilitate read across.
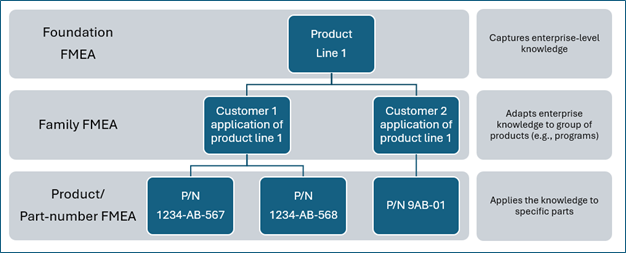
Defining what your foundations and families FMEA are going to be is highly dependent on your organization, its products and processes, and the tool or software you are using in your FMEA process. As a general rule, a foundation FMEA should be generic enough to capture product and process knowledge accumulated throughout the product’s lifecycle. Try to keep the number of foundations FMEAs manageable so they can be easy to maintain.
Examples of foundation Design FMEAs could include: a function or a generic product design (i.e. a half-shaft). Example for a family DFMEA could be a half shaft with a specific type of joint and tube type/length. Design foundations can also be defined for different levels of integration in an assembly, sub-assembly or a component, if they add value to your FMEA process. Examples of a foundation for process FMEA could be an assembly process/line, a work center, an operation, or even an activity/task. Whichever way your foundations are defined, they should be able to be used as the starting point for all new families and part number-specific FMEAs, therefore ensuring that all accumulated knowledge (i.e. lessons learned) is transferred to new products or programs.
Now that you understand and have your FMEAs structured this way, let’s walk through how enabling read across works.
When a lesson learned is identified (from structured problem solving, warranty investigations, new knowledge on new programs, continuous improvements projects, etc.), you will review it and determine the scope of the read across. Let us assume that we recently learned that a part escaped our process without a key machining feature. The team later determined that it was caused by a worn-out tool and implemented a tool cycle counter with limits to preventively change the tool before it wears out, and a camera to detect the presence of the missed feature. Because this condition could potentially occur anywhere this product is machined, the team documented both actions in the optimization section (step 6) of the foundation process FMEA for that product line.
Because of the hierarchical structure set up the action in step 6 will be added to all children (family) and grandchildren (product) FMEA, triggering actions to evaluate and implement the new controls or similar controls for the identified lesson learned anywhere the product is machined in the organization.
In time, you will need to verify the implementation of these actions to confirm read across. This is where your internal audit program can help. A Quality Management System requires internal audits, and particularly an Automotive QMS requires organizations to perform manufacturing process audits. If you plan the process audit to be performed utilizing reverse process FMEA, you can verify read across, perform the process audit, and perform reverse process FMEA all concurrently. This avoids duplicating efforts (increases efficiency), plus ensures a more effective and robust approach to the verification of read across and a value-added process audit.
Sustaining FMEAs and ensuring read across are two distinct areas that can challenge any QMS. Keeping up with these two requirements often lead to overly complex systems that are difficult to maintain, hard to follow, and ultimately discourage consistent use. By leveraging and enhancing the processes and requirements of your QMS in innovative ways, organizations can reduce complexity, save time, and create a more effective and predictable system for the lessons learned read across.
Subscribe to our newsletter.